NOESY and EXSY
1H-1H NEOSY (Nuclear Overhauser Effect SpectroscopY) and EXSY (EXchange SpectroscopY) are the same experiment used for different purposes. NOESY is useful for determining which signals arise from protons that are close to each other in space even if they are not bonded. A NOESY spectrum yields through space correlations via spin-lattice relaxation. NOESY also detects chemical and conformational exchange. It is called EXSY when used for this purpose.
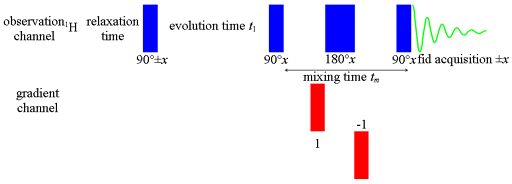
We use regular or the gradient enhanced NOESY pulse sequence (fig. 1) as they give very similar results. Just leave out the gradients for the regular sequence. The mixing time (tm) should be between half T1 and T1 to achieve good sensitivity for spectral assignment by NOESY. However, for quantitative NOESY, short mixing times and long relaxation delays are required. The strength of the NOE signal is proportional to the inverse sixth power of the distance between the atoms, I µ 1/r6. Comparison of the cross-peak integrals in a quantitative NOESY is used as a measure of the distance between the protons. A quantitative EXSY is used to calculate the rate of exchange of two or more nuclei. Sensitivity of NOESY is increased by increasing the longitudinal relaxation time either by choosing a low-viscosity solvent such as acetone-d6 and/or removing dissolved oxygen from the sample.
Fig. 1. Pulse sequence for gradient NOESY
The NOESY spectrum as shown in fig. 2 for ethylbenzene (fig. 3) contains a diagonal and cross peaks. The diagonal consists of the 1D spectrum. The cross peaks signals arising from protons that are close in space. All the desired signals are pure phase, either positive or negative. NOE signals are positive for small molecules in low-viscosity solvents and negative for very large molecules or very viscous solvents relative to a negative diagonal. By convention one should plot the diagonal as negative but most people plot the diagonal positively so the cross-peaks appear in the opposite sign to the convention: negative for small molecules and positive for large molecules. Here, we plot the diagonal as positive.
Fig. 2. 2D NOESY spectrum of ethylbenzene
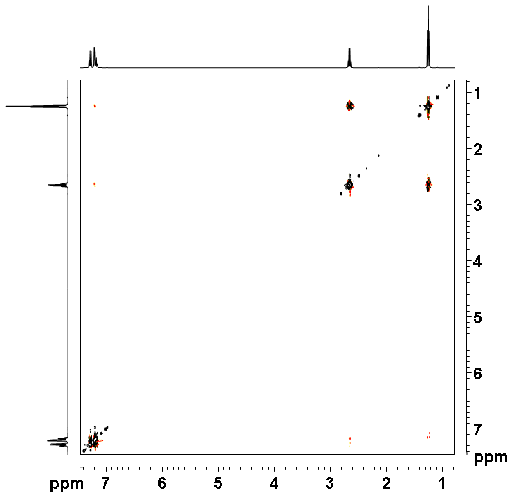
Fig. 3. Structure of ethylbenzene
Ethylbenzene does not show strong NOE correlations. The correlation between H1' and H2' at 2.65 and 1.24 ppm is mostly COSY type. A much weaker through-space correlation appears between H1' and H2 at 2.65 and 7.20 ppm. For intermediate sized molecules, the NOE signals might be of zero intensity in which case a ROESY spectrum will be more sensitive as ROE signals are always positive. Exchange peaks are negative by convention but would appear positive as plotted here. COSY-type artifacts (fig. 4) appear in correlations that are also bonded. The COSY signal is dispersive and anti-phase and may be combined with a pure phased NOESY signal. In addition, artifacts (undesired signals, fig. 5) also appear in the spectrum as vertical streaks (interference and f1 noise) and along the inverted 'V'. These artifacts are rarely in phase with the desired signals and appear in specific locations.
Fig. 4. Dispersive COSY-type cross-peak in the NOESY spectrum of ethylbenzene
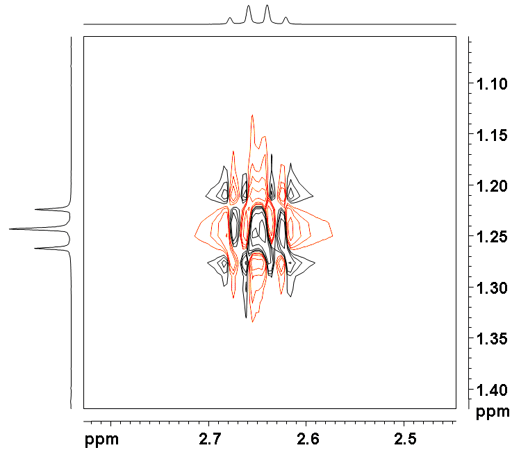
Fig. 5. Artifacts in the NOESY spectrum of ethylbenzene
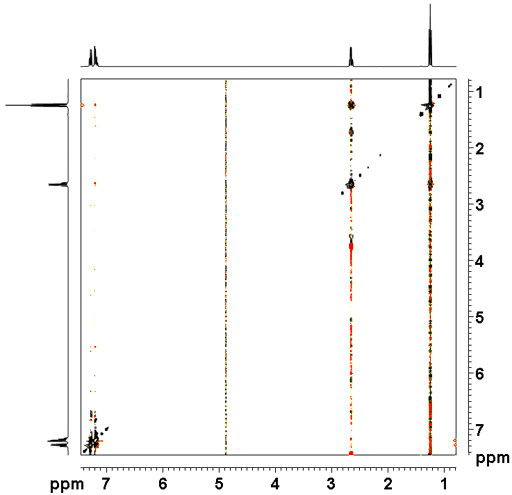
For example, in 12,14-ditbutylbenzo[g]chrysene (fig. 6), only a partial analysis of the regular 1H-NMR spectrum is possible. NOESY (fig. 7) provides extra information about the connectivity allowing a full assignment based on through-space correlations. As a result, the spectrum displays correlations to the tbutyls and between ring systems allowing a full assignment of the proton spectrum.
Fig. 6. Structure of 12,14-ditbutylbenzo[g]chrysene
![12,14-ditbutylbenzo[g]chrysene](noesy_files/dtbbgc.gif)
Fig. 7. 2D NOESY spectrum of 12,14-ditbutylbenzo[g]chrysene
![NOESY of 12,14-ditbutylbenzo[g]chrysene](noesy_files/noesydtbbgc.gif)
The aromatic region of the spectrum (fig. 8) shows three bond correlations that include a COSY type component in dispersive phase and negative pure phase signals for correlations between ring systems. These can be used to determine which protons are neighbors. For example the proton at 8.17 ppm is next to the proton at 7.34 ppm on the same ring, a fact that could not be easily determined from the 1D spectrum. The proton at 8.56 is on the ring next to the signal at 8.32 ppm, a fact that could not be determined from the 1D or the COSY spectrum. This shows the proton at 8.56 ppm is in a group of four protons next to a group of two protons. The only case in this molecule is the correlation between H8 and 9 which are now known to be at 8.56 and 8.32 ppm respectively.
Fig. 8. Aromaric region of the 2D NOESY spectrum of 12,14-ditbutylbenzo[g]chrysene
![Aromatic region of NOESY of 12,14-ditbutylbenzo[g]chrysene](noesy_files/aromaticdtbbgc.gif)
Continuing the connectivity, we can assign H10 as 7.76 ppm H11 as 7.60 ppm and H13 as 7.86 ppm. In the opposite direction, H7 is at 7.59 ppm , H6 at 7.55 ppm, H5 at 8.62 ppm, H4 at 8.54 ppm, H3 at 7.44 ppm, H2 at 7.34 ppm and H1 at 8.17 ppm. Fig. 9 shows the NOESY connectivity colored according their assignemnt to each ring (fig. 10).
Fig. 9. Aromaric region of the 2D NOESY spectrum of 12,14-ditbutylbenzo[g]chrysene showing connectivity and separation into four color-coded proton groups
![Aromatic region of NOESY of 12,14-ditbutylbenzo[g]chrysene in color](noesy_files/coloraromatic.gif)
Fig. 10. Structure of 12,14-ditbutylbenzo[g]chrysene showing color conding of rings
![Color coded structure of 12,14-ditbutylbenzo[g]chrysene](noesy_files/dtbbgccolor.gif)
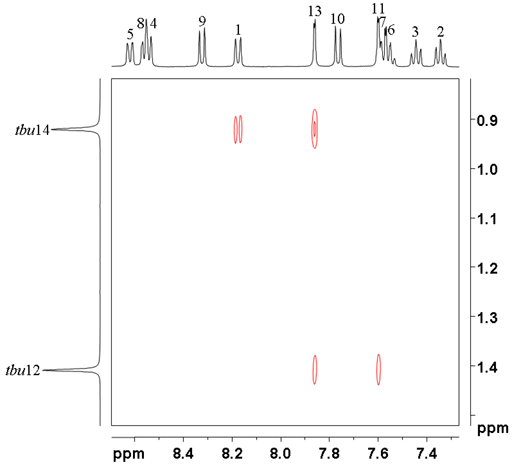
All that remains is to assign the tbutyls. In the NOESY, tbu14 at 0.92 ppm correlates with H1 and 13 while tbu12 at 1.40 ppm correlates with H11 and 13 (fig. 11) confirming the provisional assignment of the 1H spectrum.
Fig. 11. tButyl regrion of the NOESY of 12,14-ditbutylbenzo[g]chrysene