Identify Your Compound
Compound identification
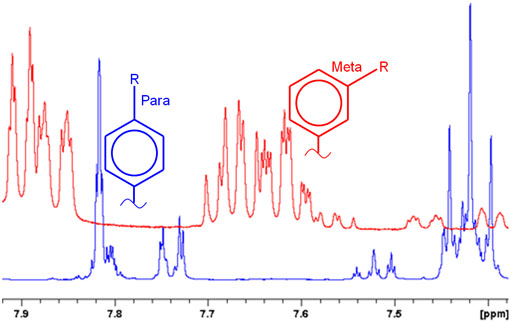
NMR's ability to differentiate subtle differences in molecular configuration makes it an ideal technique for identifying your compound
Use Our NMR service for all your NMR needs.
Molecular configuration
NMR spectroscopy is best suited to determining the connections between atoms within a molecule and hence the full molecular structure. Using the spectral parameters, it is possible to assign the spectrum and deduce the properties of the material.
Fig. 1. Very different NMR spectra arise from a small change in configuration such as the difference between meta and para substitution
HPLC & GC-MS don't work? Try NMR
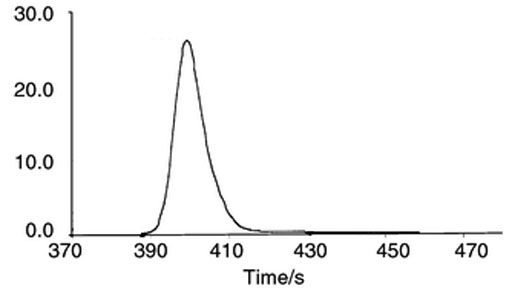
Unless there is a large difference in moleclar weight or interaction with the solvent, HPLC and GC-MS will not differentiate between different compounds. This is where NMR comes to the rescue especially for isomers and minor changes in substitution.
Fig. 2. HPLC cannot separate similar compounds
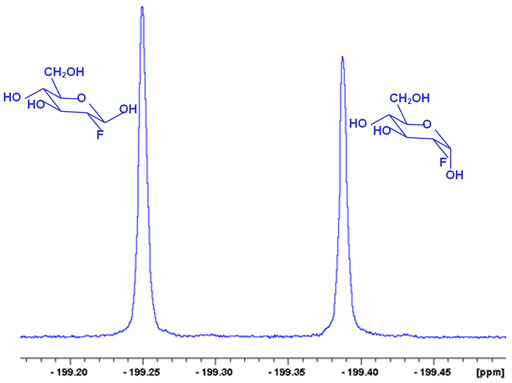
In the example below, two very similar isomers (anomers) of 2-fluoro-2-deoxyglucose are clearly separated by 19F NMR.
Fig. 3. NMR can!