(209Bi) Bismuth NMR
Use our NMR service that provides 209Bi NMR and many other NMR techniques.
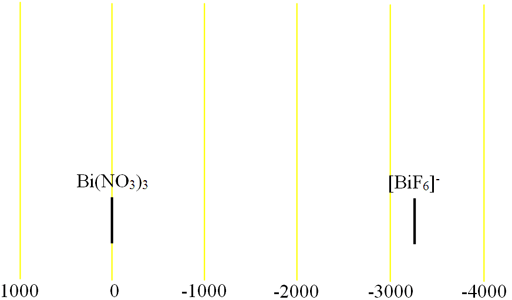
Bismuth (Bi) has one high sensitivity NMR active nucleus, 209Bi (fig. 1). It is quadrupolar and yields very broad signals even in the most symmetric of environments over a very wide chemical shift range. Only Bi3+ and [BiF6]- have been observed by high resolution NMR so chemical shift information (fig. 2) is very limited.
Fig. 1. 209Bi-NMR spectrum of Bi(NO3)3 in nitric acid/D2O
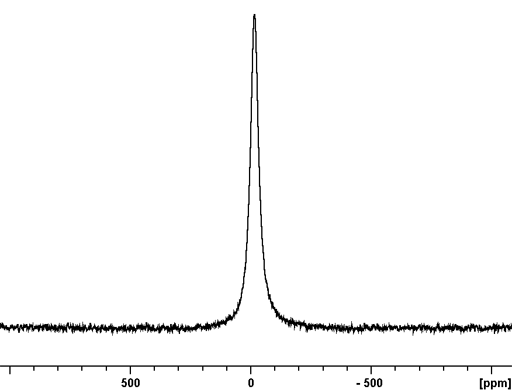
Fig. 2. Chemical shift ranges for bismuth NMR
Properties of 209Bi
| Property | Value |
|---|---|
| Spin | 9/2 |
| Natural abundance | 100% |
| Chemical shift range | 3300 ppm, from 0 to -3300 |
| Frequency ratio (Ξ) | 16.069288% |
| Reference compound | sat. Bi(NO3)3 in conc. HNO3 + 50% D2O |
| Linewidth of reference | 2978 Hz |
| T1 of reference | 0.000052 s |
| Receptivity rel. to 1H at natural abundance | 0.144 |
| Receptivity rel. to 1H when enriched | 0.144 |
| Receptivity rel. to 13C at natural abundance | 848 |
| Receptivity rel. to 13C when enriched | 848 |
| Linewidth parameter | 200 fm4 |
Safety note
Some of the materials mentioned here are very dangerous. Ask a qualified chemist for advice before handling them. Qualified chemists should check the relevant safety literature before handling or giving advice about unfamiliar substances. NMR solvents are toxic and most are flammable. Specifically, bismuth salts are toxic in large quantities besides any toxicity arising from the anion. 209Bi is very slightly radioactive but not to an extent that it can be dangerous.
References
- K. Morgan, B. G. Sayer, G. J. Schrobilgen, "Bismuth NMR spectroscopy: 209Bi and 19F high-resolution NMR spectra of the hexafluorobismuthate(V) Ion", J. Magn. Reson., 1, 139-142 (1969).