(55Mn) Manganese NMR
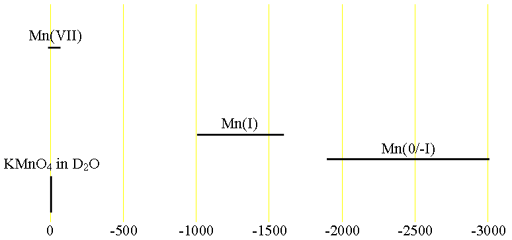
55Manganese (55Mn) is the only NMR active nucleus of manganese. It is a high sensitivity nucleus that yields moderately broad lines (fig. 1) in symmetric environments over a very wide chemical shift range. For larger complexes and molecules, the signals become too broad to observe with a high-resolution NMR spectrometer. Oxidation states, (-I), (O), (I) and (VII) of manganese are diamagnetic and yield observable signals for small complexes. Other oxidation states are paramagnetic and cannot be observed by high-resolution NMR. Manganese NMR is used for studying small complexes of manganese and manganese binding. There is little information available about chemical shifts (fig. 2).
Fig. 1. 55Mn-NMR spectrum of KMnO4 (sat.) in D2O
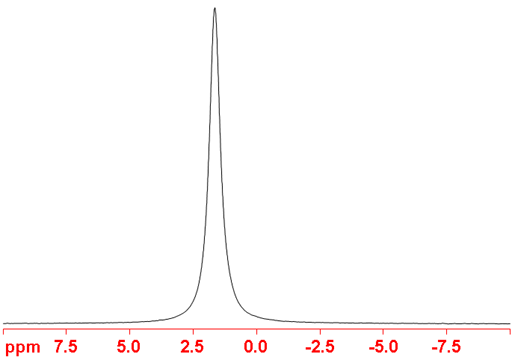
Fig. 2. Chemical shift ranges for manganese NMR
Properties of 55Mn
| Property | Value |
|---|---|
| Spin | 5/2 |
| Natural abundance | 100% |
| Chemical shift range | 3000 ppm, from -3000 to 0 |
| Frequency ratio (Ξ) | 24.789218% |
| Reference compound | 0.82 m KMnO4 in D2O |
| Linewidth of reference | 5.6 Hz |
| T1 of reference | 0.072 s |
| Receptivity rel. to 1H at natural abundance | 0.179 |
| Receptivity rel. to 1H when enriched | 0.179 |
| Receptivity rel. to 13C at natural abundance | 1050 |
| Receptivity rel. to 13C when enriched | 1050 |
| Linewidth parameter | 350 fm4 |
Safety note
Some of the materials mentioned here are very dangerous. Ask a qualified chemist for advice before handling them. Qualified chemists should check the relevant safety literature before handling or giving advice about unfamiliar substances. NMR solvents are toxic and most are flammable. Specifically, manganese compounds are toxic and KMnO4 is oxidizing: wear gloves.